
CDH UK Christmas Magazine 2025 Edition
The CDH Magazine 2025 Edition has been released, click the link below to take a look! The CDH Magazine Edition 6 2025

The CDH Magazine 2025 Edition has been released, click the link below to take a look! The CDH Magazine Edition 6 2025

This year our Annual General Meeting will take place online via the Zoom platform Date 30th November Time 10.00am – 11.00am To register for the

The CDH Magazine 2024 Edition has been released, click the link below to take a look! The CDH Magazine Edition 4 2024
Join in June 4 CDH has been an official awareness initiative started by CDH UK in 2010 to encourage awareness and action for Congenital Diaphragmatic

Collaborating to improve outcomes The main CDH research event is held every two years. This time it was the turn of France and the beautiful

The CDH Magazine 2023 Edition has been released, click the link below to take a look! The CDH Magazine Edition 3 2023
IMPORTANT UPDATE: Due to unforeseen circumstances beyond our control the CDH UK Committee has decided to change the in-person meeting to an online virtual meeting,
Join in June 4 CDH has been an official awareness initiative started by CDH UK in 2010 to encourage awareness and action for Congenital Diaphragmatic
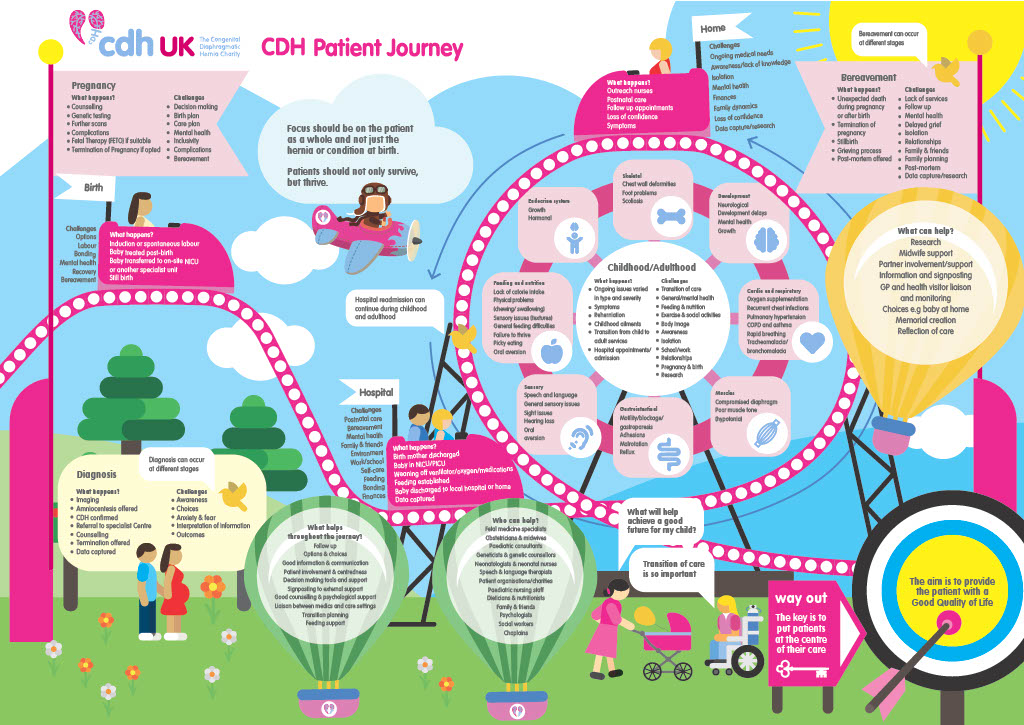
We are very proud and excited to have had The CDH Patient Perspective Journey published in Frontiers in Pediatrics (neonatology section) in February 2023. We

The CDH Magazine 2022 Edition has been released, click the link below to take a look! The CDH Magazine Edition 3 2022
We are deeply saddened by the death of our much loved Her Majesty The Queen and our heartfelt condolences are offered to the Royal Family.

June is the month to raise extra awareness for Congenital Diaphragmatic Hernia (CDH) and June 28th is awareness day. Throughout June there will be activities

CDH2022 is a follow on from previous symposia that is this time being held in Glasgow, Scotland. Bringing together Medical professionals, Patients Organisations and Researchers,

The CDH Magazine 2021 Edition has been released, click the link below to take a look! The CDH Magazine Edition 2 2021

New guidelines to improve pregnancy trials will pave way for novel therapies for women and babies. A new grading system to monitor complications during clinical

If you love a quiz, you will love this CDH Christmas Cracker Quiz! Even if you don’t you should still join in and help raise

Chairperson of CDH UK Beverley Power, along with other Rare Disease experts wrote to The Lancet to voice their concerns of a no deal Brexit
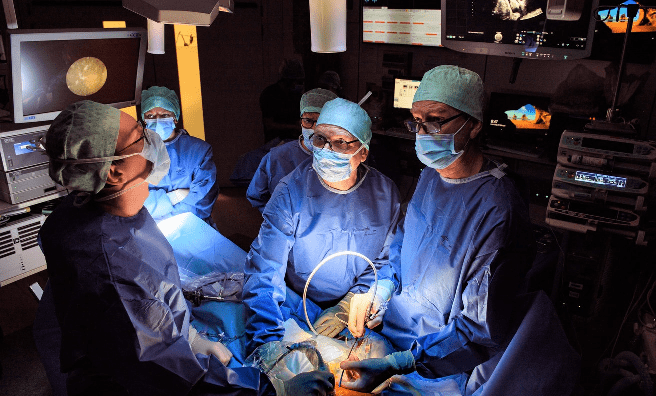
Many years ago a team based in Leuven, led by Professor Jan Deprest, set out on an incredible journey with the help of a little

We have set up a nice little virtual challenge for you that encourages you to do 5K Your Way 4 CDH UK and to acknowledge

We have renamed our Newsletter The CDH Magazine! It will still feature everything that the newsletter featured but now has the ability to feature even
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.